Health News
33 seconds ago
Wanna flex your green thumb this season? According to a professional gardener, you should have these 11 tools on hand
I write about home and kitchen products for a living, so if you’re looking for a new vacuum or air fryer, I’m your gal. But I’ll be the first to admit that when it comes to gardening,…
Health News
12 hours ago
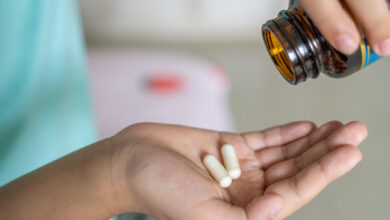
Melatonin bottles are easy for young kids to open. New guidelines could change that.
After thousands of children wound up in U.S. emergency rooms after accidentally ingesting melatonin, a major supplement industry group has issued new safety guidelines, which include asking makers of the popular sleep aid to use child-deterrent packaging.…
Health News
1 day ago
These bestselling satin pillowcases have nearly 225,000 five-star reviews — and they’re on sale for $6 a pair
With so much of our lives spent in dreamland, having great bedding is a must. So imagine our delight when we found a set of pillowcases that will not only make your bedroom look and feel more…
Health News
2 days ago
How to stay safe during heavy rain, lightning and tornadoes
Storms sweeping across the central U.S. beginning on April 15 could inundate as many as 40 million Americans with large hail, high-speed winds and even tornadoes, meteorologists warned Monday. Weather — from heat to cold to tornadoes…
Health News
2 days ago
Shoppers are ‘ridiculously thankful’ for this No. 1 bestselling $25 anti-hair-loss duo — now nearly 40% off
Shed happens. We typically lose about 50 to 100 strands of hair per day, according to the American Academy of Dermatology Association. That might not amount to much, considering we have roughly 100,000 hairs on our heads…